Inflammation
Inflammation Definition:
Inflammation is a response of vascularized tissues to infection and damaged tissues that brings cells and molecules of host defense from circulation to the sites where they needed, in order to eliminate the offending agents.
Typical Inflammatory reaction develops through a series of sequential steps.
Sequence of events in inflammatory reaction
- Recognition of the noxious agent
- Recruitment of leukocytes and plasma protein into the tissues
- Removal of the stimulus for inflammation is accomplished mainly by phagocytic cells, which ingest and destroy microbes and dead cells.
- Regulation of the response is important for terminating the reaction when it has accomplished its purpose.
- Repair: In this process, the injured tissue is replaced through the regeneration of surviving cells and the filling of residual defects with connective tissue (scarring).
Properties of Inflammation
- Components of the inflammatory response: Blood vessels, Leukocytes
- Harmful consequences of inflammation: Accompanied by local tissue damage (e.g., against self-tissues in autoimmune diseases).
- Local and systemic inflammation
- Mediators of inflammation
- Acute and chronic inflammation
- Termination of inflammation and initiation of tissue repair
Advantages of inflammation
- Inflammation is a protective response
- Serves to destroy, dilute or wall off the injurious agents (microbes, toxins).
- Remove necrotic cells and tissues
- Prepare any damaged tissue for repair and help in healing.
Disadvantages of inflammation
- Local tissue damage and associated sign and symptoms (pain and functional impairment).
- May leads to chronic diseases
- Rheumatoid arthritis
- Atherosclerosis
- Lung fibrosis
- Hypersensitivity reaction
- Repair by fibrosis may lead to disfiguring scar.
Disease caused by inflammatory reactions
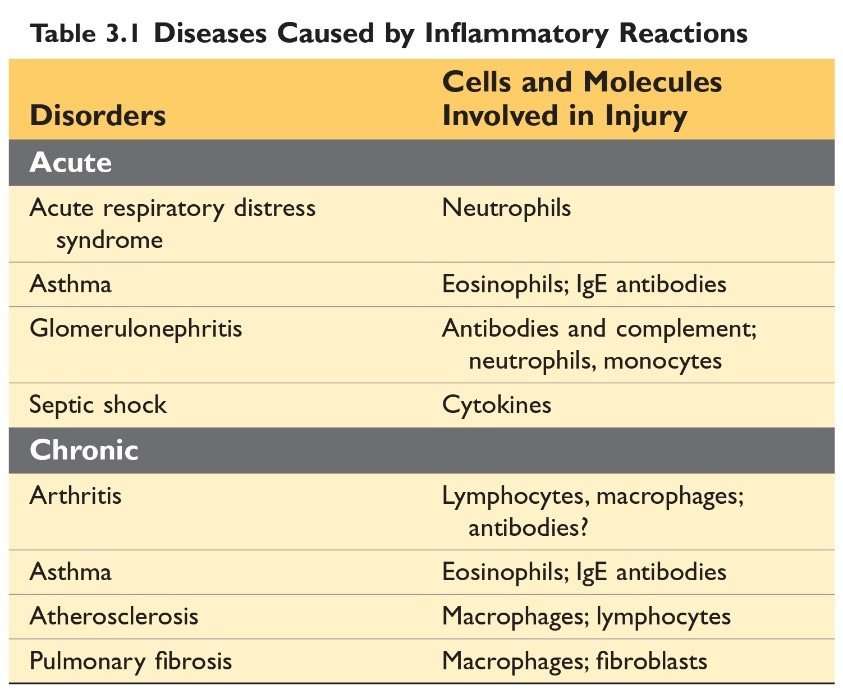
Source: Robbin’s 10th Edition (Page 73)
Difference between Acute inflammation and chronic inflammation
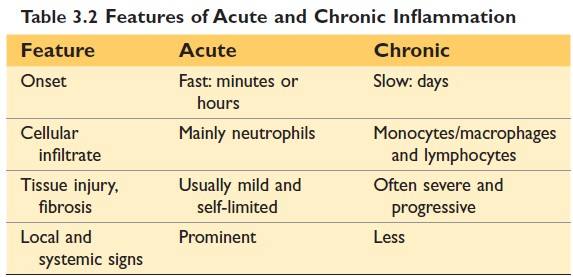
(Source Robbin's 10th Edition)
Cardinal signs of inflammation:
- Rubor (Redness): Due to vessel dilation and increased blood flow.
- Calor (Heat): Due to vessel dilation and increased blood flow)
- Dolor (Pain): Due to a combination of pressure on nerve endings from swelling and direct of chemical mediators.
- Tumor (Swelling): Due to accumulation of fluid.
- Functio laesa (Loss of function)
Causes of inflammation
- Infection (bacteria, viral, fungal infection & parasitic infestation) and microbial toxins.
- Tissue necrosis from any cause
-
- Ischemia (as in a myocardial infarct), trauma, and
- Physical and chemical injury (e.g. Thermal injury, as in burns of frostbite, irradiation)
- Exposure to some environmental chemicals.
- Foreign bodies
- Immune reactions
Types of inflammation:
- Acute inflammation
- Chronic inflammation
Acute inflammation:
Acute inflammation is a rapid response (within minutes or hours) to injurious agents and lasting for several hours or a few days and characterized by an exudation of fluid and plasma proteins, and emigration of leukocytes, principally neutrophils.
Events or components of inflammatory response
- Dilation of small vessels leading to an increase in blood flow.
- Increased permeability of the microvasculature with leakage of plasma proteins and leukocytes to from the circulation and
- Emigration of leukocytes from microcirculation, their accumulation in the focus of injury, and their activation to eliminate the offending agent.
Some Terms
| Exudate: An exudate is an extravascular fluid that has a high protein concentration and contains cellular debris. The escape of fluid, proteins, and blood cells from the vascular system into the interstitial tissue or body cavities is known as exudation. |
| Transudate: A transudate is a fluid with low protein content (most of which is albumin), little or no cellular material, and low specific gravity. It is essentially an ultrafiltrate of blood plasma that is produced as a result of osmotic or hydrostatic imbalance across the vessel wall without an increase in vascular permeability. |
| Edema: Edema denotes an excess of fluid in the interstitial tissue or serous cavities; it can be either an Exudate or a Transudate. |
| Pus: Pus, a purulent exudate, is an inflammatory exudate rich in leukocytes (mostly neutrophils), the debris of dead cells, and in many cases, microbes. |
Reactions of Blood Vessels to Inflammation:
Changes in the flow of blood and permeability of vessels, and maximize the movement of plasma proteins and leukocytes out of the circulation and into the site of the infection or injury.
- Increased Vascular Permeability
- Responses of Lymphatics Vessels and Lymph nodes
Increased Vascular Permeability
It is a hallmark of acute inflammation where there is an Escape of fluid and plasma protein (exudation)
Mechanism of Increased Vascular Permeability
- Contraction of endothelial cell
- Direct endothelial injury
1) Contraction of endothelial cell
Contraction of endothelial cells results in the opening of inter-endothelial gaps.
- It is the most common mechanism of vascular leakage.
- Elicited by histamine, bradykinin, leukotrienes, and other chemical mediators.
It is called an immediate transient response because it occurs rapidly after exposure to the mediator and is usually short-lived (15-30 minutes).
Vascular leakage begins after a delay of 2 to 12 hours and lasts for several hours or even days; this delayed prolonged leakage may be caused by the contraction of endothelial cells or mild endothelial damage.
Caused by some forms of mild injury (e.g. after burns, irradiation or ultraviolet radiation, and exposure to certain bacterial toxins).
Sunburn is a classic example of the damage that results in late-appearing vascular leakage. Often the immediate and delayed responses occur along a continuum.
2) Endothelial injury
Endothelial injury resulting in endothelial cell necrosis and detachment.
Example: Physical injuries, like in thermal burns and microbial toxins. Neutrophils that adhere to the endothelium during inflammation may also injure endothelial cells & thus amplify the reaction.
In most instances leakage starts immediately after injury and is sustained for several hours until the damaged vessels are thrombosed or repaired.
Responses of Lymphatics Vessels and Lymph nodes
In addition to blood vessels, lymphatic vessels also participate in acute inflammation.
In inflammation, lymph flow is increased and helps drain edema fluid that accumulates because of increased vascular permeability. In addition to fluid, leukocytes and cell debris, as well as microbes, may find their way into the lymph.
Nice to Know:
The lymphatics may become secondarily inflamed (lymphangitis), as may the draining lymph nodes (lymphadenitis). Inflamed lymph nodes are often enlarged because of hyperplasia of the lymphoid follicles and increased numbers of lymphocytes and macrophages.
This constellation of pathologic changes is termed reactive, or inflammatory, lymphadenitis.
Cellular events of inflammation
(Leukocyte Recruitment to Sites of Inflammation)
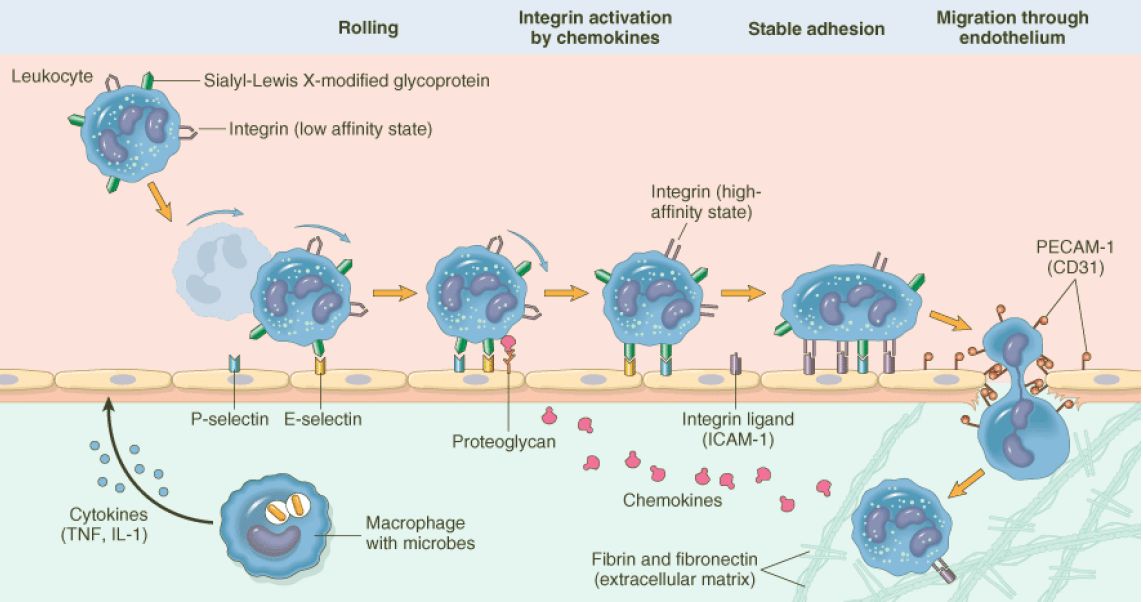
Leukocytes' extravasation and transmigration
- In the lumen:
- Margination
- Rolling and
- Adhesion to endothelium
- Transmigration across the endothelium and vessel wall (Diapedesis)
- Chemotaxis
- Phagocytosis
Margination: Accumulation of leukocytes along the endothelium.
Rolling: Individuals and rows of leukocytes tumble slowly along the endothelium.
Leucocytes adhesion to endothelium: Regulated by the bindings of adhesion molecules on the leukocytes and endothelial surface.
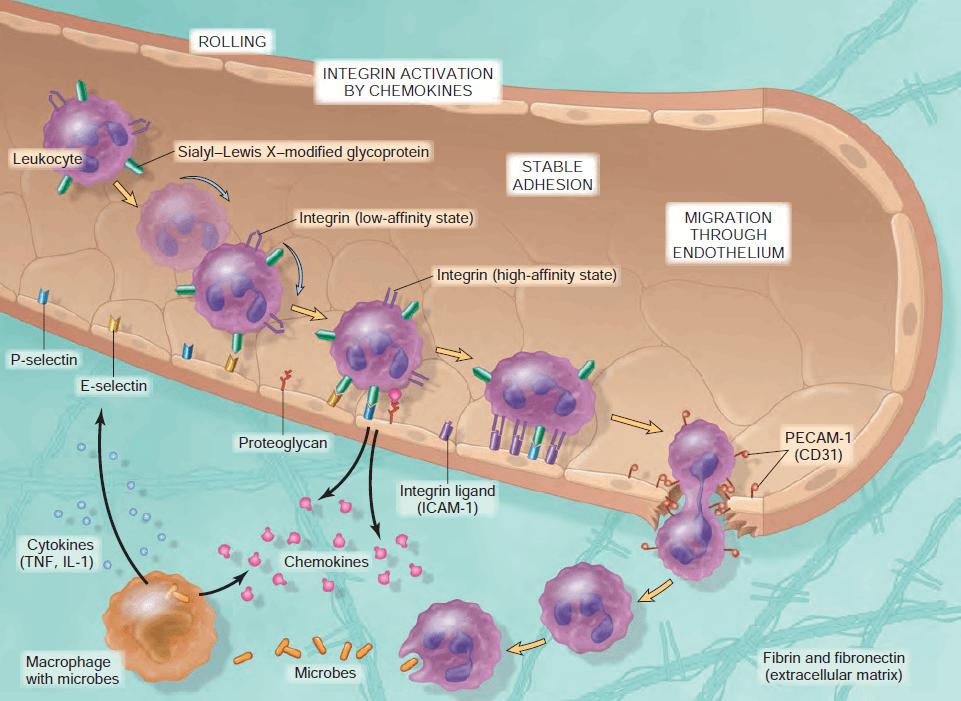
Process of leukocyte migration from blood vessels
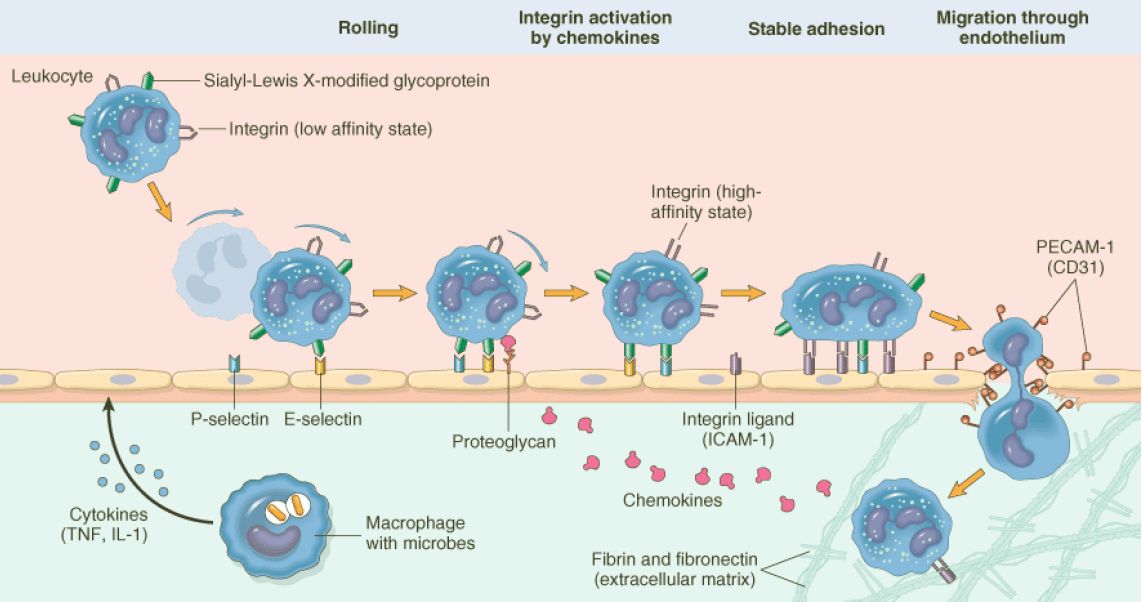
Figure: Process of leukocyte migration from blood vessels
Source: Robbin’s 7th Edition
Adhesion molecule:
- Selectins: Expressed on leukocytes (L-selectin), on endothelium (E-selectin), and one in platelets and on endothelium (P-selectin). The ligands for selectins are sialylated oligosaccharides bound to mucin-like glycoproteins.
- Integrins: Ligands are ICAM-1 (Intercellular adhesion molecule-1), VCAM-1 (Vascular cell adhesion molecule 1)
- Immunoglobulin family: CD31 also called PECAM 1 (Platelet endothelial cell adhesion molecule)
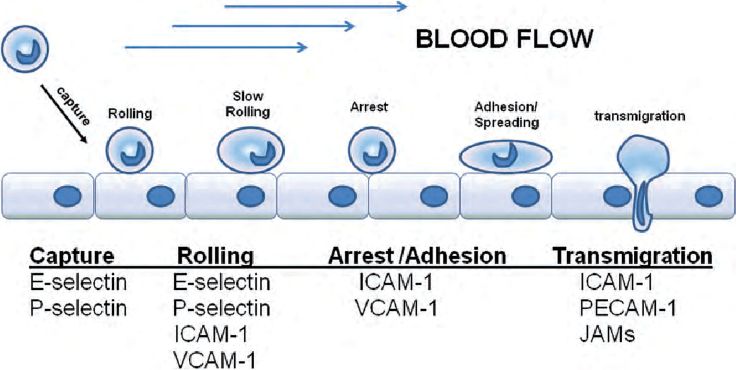
Source: Internet (Function of leukocyte adhesion molecules)
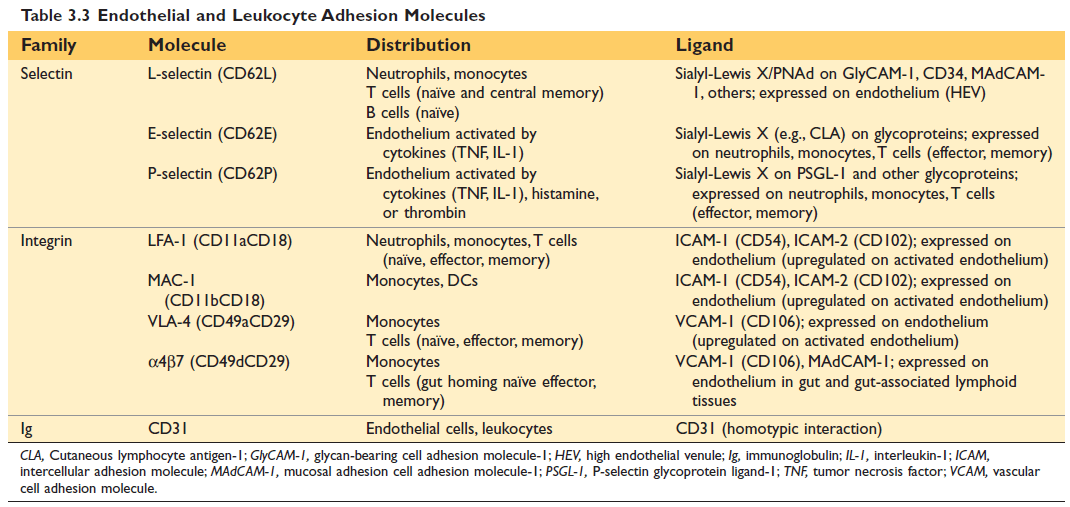
Figure: Endothelial and Leukocyte Adhesion Molecules (10th Edition Robbin's)

Figure: Leukocyte Recruitment to the site of inflammation Robbin's
Diapedesis (Transmigration):
Migration of the leukocytes through intact endothelium is called transmigration or diapedesis.
Leukocytes pierce the basement membrane, probably by secreting collagenases and enter the extravascular space.
Chemotaxis:
After exiting the circulation, leukocytes move in the tissue toward the site of injury by a process called chemotaxis, which is defined as locomotion along a chemical gradient.
Chemotactic factor or Chemoattractans: (Chemotaxis of Leukocytes)
- Exogenous:
- Bacterial products: Peptides (N-formylmethionine terminal amino acid), and some lipids.
- Endogenous:
- Components of the Complement system: C5a
- Product of lipoxygenase pathway: LTB4 (Leukotriene B4)
- Cytokines (eg. IL-8)
Nature of leukocyte infiltrate
- Varies with the duration of the inflammatory response and the type of stimulus.
- Neutrophils predominate in the inflammatory infiltrate during the first 6 to 24 hours and are replaced by monocytes in 24 to 48 hours.
Nice To Know
- In certain infections—for example, those produced by Pseudomonas bacteria—the cellular infiltrate is dominated by continuously recruited neutrophils for several days;
- In viral infections, lymphocytes may be the first cells to arrive;
- Some hypersensitivity reactions are dominated by activated lymphocytes, macrophages, and plasma cells (reflecting the immune response); and
- In helminthic infections and allergic reactions, eosinophils may be the main cell type.
Reasons account for the early appearance of neutrophils:
- They are more numerous in the blood,
- They respond more rapidly to chemokines, &
- They may attach more firmly to the adhesion molecules that are rapidly induced on endothelial cells, such as P-selectin and E-selectins.
- After entering tissues, neutrophils are short-lived, they undergo apoptosis and disappear after 24 to 48 hours.
Phagocytosis:
It is the process of engulfment and ingestion of bacteria or other foreign bodies by phagocytes (Neutrophils, macrophages).
Recognition of microbes or dead cells induces several responses in leukocytes that are collectively called leukocyte activation.
Steps of Phagocytosis Involves 3 sequential steps
- Recognition and attachment
- Engulfment (with subsequent formation of a phagocytic vacuole)
- Killing or degradation
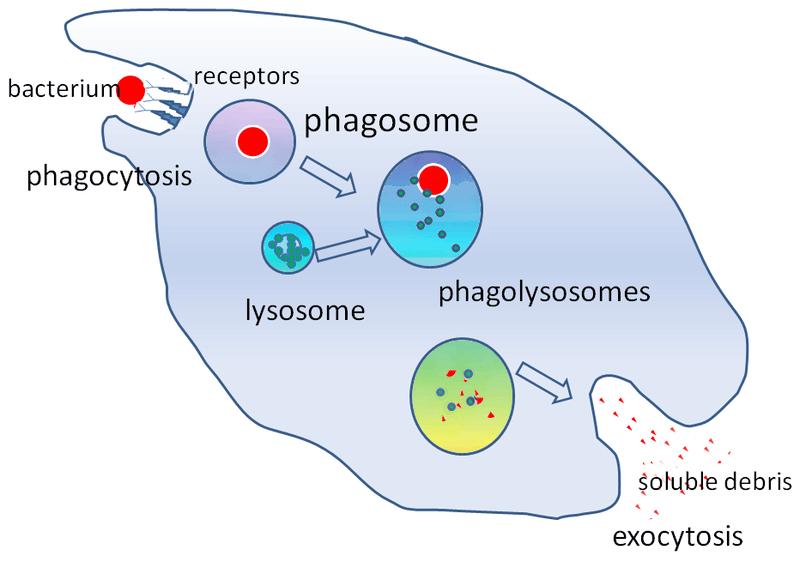
Figure: Steps of phagocytosis
Recognition and attachment:
The efficiency of phagocytosis is greatly enhanced when microbes are opsonized by specific proteins (opsonin).
Major opsonins are
- Fc fragment of IgG antibodies,
- The C3b and
- Certain plasma lectins, notably mannan-binding lectin.
Engulfment:
During engulfment, the extension of the cytoplasm (pseudopods) flows around the particle to be engulfed, resulting in a complete closure of the particle within a phagosome created by the plasma membrane of the cell. The phagocytic vacuole then fuses with the lysosome (phagolysosome), resulting release of the granule content into the phagolysosome.
Killing and degradation:
The killing of microbes is accomplished by
- Reactive oxygen species (ROS), also called reactive oxygen intermediates and
- Reactive nitrogen species, mainly derived from nitric oxide (NO), and
- Lysosomal enzymes
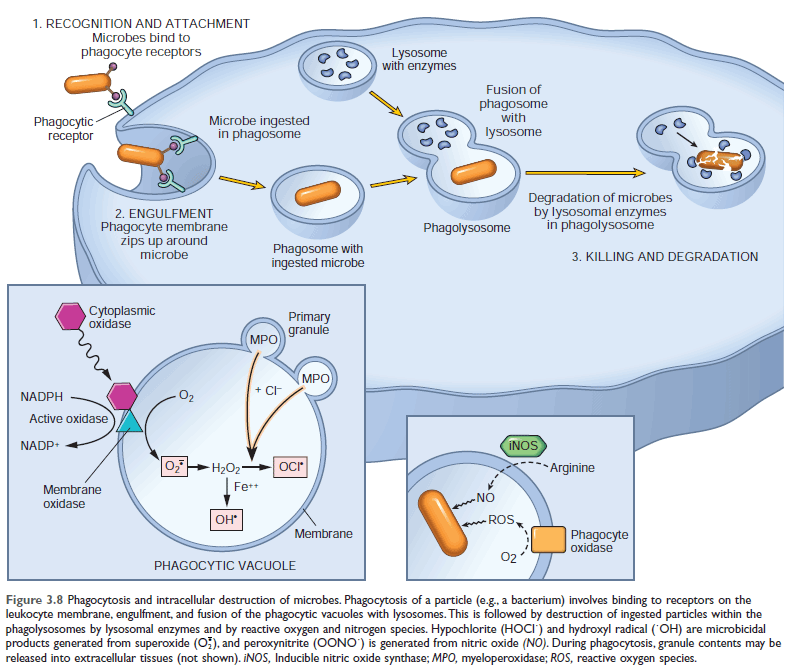
Showing: 1. Phagocytosis process & 2. Production of microbicidal reactive oxygen intermediates within the phagocytic vacuole (Reaction within phagocytic vacuole)
Reactive Oxygen Species:
ROS are produced by the rapid assembly and activation of a multicomponent oxidase, NADPH oxidase (also called phagocyte oxidase), which oxidizes reduced nicotinamide-adenine dinucleotide phosphate (NADPH) and, in the process, reduces oxygen to superoxide anion (O2-).
In neutrophils, this oxidative reaction is triggered by activating signals accompanying phagocytosis and is called the respiratory burst.
(ROS are produced within the phagolysosome, where they can act on ingested particles without damaging the host cell.)
Superoxide (O2-) is then converted into H2O2, mostly by spontaneous dismutation.
H2O2 is not able to efficiently kill microbes by itself. However, the azurophilic granules of neutrophils contain the enzyme myeloperoxidase (MPO), which, in the presence of a halide such as Cl-, converts H2O2 to hypochlorite (HOCl?).
The H2O2-MPO-halide system is the most potent bactericidal system of neutrophils.
H2O2 is also converted to hydroxyl radical (OH), another powerful destructive agent.
Brainstorming:
|
Is there any problem, if a person is deficient of phagocyte oxidase?
The deficiency of Phagocyte oxidase components (i.e., NADPH oxidase) causes Chronic Granulomatous Disease (CGD) which is an Inherited immunodeficiency disease. |
Nitric Oxide:
NO, a soluble gas produced from arginine by the action of nitric oxide synthase (NOS), also participates in the microbial killing.
In macrophages, NO reacts with superoxide (O2-) to generate the highly reactive free radical peroxynitrite
(ONOO−). These nitrogen-derived free radicals, similar to ROS, attack, and damage the lipids, proteins, and nucleic acids of microbes.
Nitric oxide synthase:
3 types of NOS:
- Endothelial (eNOS),
- Neuronal (nNOS),
- Inducible (iNOS): Play role in inflammation.
Other functions of Nitric oxide:
- Act as a neurotransmitter (NO produced by nNOS)
- Relaxes vascular smooth muscle and promotes vasodilation (NO produced by eNOS)
Lysosomal Enzymes and Other Lysosomal Proteins
- Role: Contributes to microbial killing and when released, may cause tissue damage also
- Source: Neutrophils and monocytes
Neutrophilic granules:
- The smaller specific (or secondary) granules
- The larger azurophil (or primary) granules
The smaller specific (or secondary) granules contain lysozyme, collagenase, gelatinase, lactoferrin, plasminogen activator, histaminase, and alkaline phosphatase.
The larger azurophil (or primary) granules contain MPO, bactericidal proteins (lysozyme, defensins), acid hydrolases, and a variety of neutral proteases (elastase, cathepsin G, nonspecific collagenases, proteinase
Neutralization of lysosomal enzymes
Destructive effects of lysosomal enzymes (proteases) controlled by antiproteases in serum and tissue fluid.
Example of antiproteases:
- α1 - antitrypsin: Inhibit neutrophil elastase
- α2 -Macroglobulin
Nice to know:
Alpha-1 antitrypsin deficiency (AATD) is an increased risk of having Chronic Obstructive Pulmonary Disease (COPD); which includes Chronic bronchitis & Emphysema
Degradation and clean-up:
After killing, dead microorganisms are degraded by lysosomal acid hydrolases within the phagolysosome.
Neutrophil Extracellular Traps:
Neutrophil extracellular traps (NETs) are extracellular fibrillar networks that concentrate antimicrobial substances at sites of infection and trap microbes in fibril, helping to prevent their spread.
Just To Know:
Culminating Chromatin decondensation – Rupture of the nuclear envelope – Release of chromatin from neutrophil – Forming Extracellular Traps
In this process, the nuclei of the nuclear envelope are lost, leading to the death of cells.
The fate of neutrophils after phagocytosis:
It undergoes apoptotic cell death and is ingested by macrophages.
The peculiarity of phagocytosis:
The contents of lysosomal granules are secreted by leukocytes into the extracellular milieu by diverse mechanisms.
- Regurgitation during feeding
- Frustrated phagocytosis
Importance: Can cause further inflammation and tissue injury.
1. Regurgitation during feeding
When phagocytic vacuole remains transiently open to the outside before complete closure of the phagolysosome and releases of lysosomal enzymes to the extracellular fluid.
2. Frustrated phagocytosis:
When the particle is too large to be engulfed or a particle is attached to a surface and cannot be engulfed and released lysosomal enzymes to the extracellular fluid.
Leukocyte-Mediated Tissue Injury:
Leukocytes are important causes of injury to normal cells and tissues under several circumstances.
- As part of a normal defense reaction against infectious microbes, when adjacent tissues suffer collateral damage. In some infections that are difficult to eradicate, such as tuberculosis and certain viral diseases, the prolonged host response contributes more to the pathology than the microbe itself.
- When the inflammatory response is inappropriately directed against host tissues, as in certain autoimmune diseases.
- When the host reacts excessively against usually harmless environmental substances, as in allergic diseases, including asthma.
The function of activated leukocytes
- Phagocytosis
- Play several other roles in host defense. Importantly, these cells, especially macrophages,
- Produce cytokines that can either amplify or limit inflammatory reactions,
- Produce growth factors that stimulate the proliferation of endothelial cells and fibroblasts and the synthesis of collagen, and enzymes that remodel connective tissues.
TERMINATION OF THE ACUTE INFLAMMATORY RESPONSE
- After the removal of the offending agent, there is reduced production of mediators, & have short half-lives, and are degraded after their release.
- Neutrophils also have short half-lives in tissues and die by apoptosis
- Anti-inflammatory lipoxins, and the liberation of anti-inflammatory cytokines, including transforming growth factor-β (TGF-β) and IL-10, from macrophages and other cells.
OUTCOME OF ACUTE INFLAMMATION:
- Resolution: Restoration of the site of inflammation to normal. Resolution involves removal of cellular debris and microbes by macrophages, and resorption of edema fluid by lymphatics.
- Abscess formation
- Healing by fibrosis
- Progress to chronic inflammation
Morphologic patterns of Acute inflammation
1. Serous inflammation:
- The outpouring of thin fluid, derived from either plasma or the secretions of mesothelial cells lining the peritoneal, pleural & pericardial cavities (effusion).
- Example: effusion, skin blister from the burn or viral infection
2. Fibrinous inflammation:
- Characterized by the formation of fibrinous exudates
Eg- pericarditis, pleuritis, meningitis.
3. Purulent (Suppurative) inflammation & Abscess:
- Characterized by the formation of pus or purulent exudates (consisting of neutrophils, necrotic cells, & edema fluid).
- Acute appendicitis, Abscess (Abscess are localized collection of purulent inflammatory tissue),
4. Ulcer: An ulcer is a local defect or excavation, of the surface of an organ or tissue that is produced by sloughing (shedding) of inflammatory necrotic tissue.
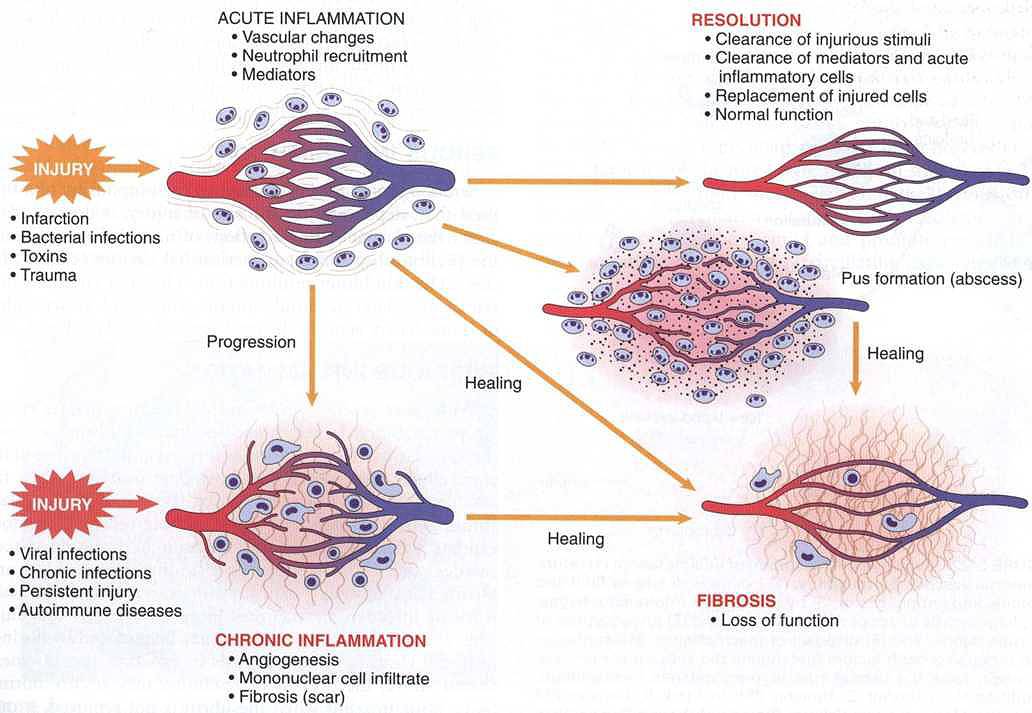
FIGURE 2.21 Outcomes of acute inflammation: resolution, healing by fibrosis, or chronic inflammation
Examples of Inflammation
| Site | Inflammation | Site | Inflammation |
| Brain | Encephalitis | Joint | Arthritis |
| Meninges | Meningitis | Subcutaneous tissue | Cellulitis |
| Nerve | Neuritis | Blood vessel | Vasculitis |
| Conjunctiva | Conjunctivitis | Artery | Arteritis |
| Cornea | Keratitis | Vein | Phlebitis |
| Uveal tract | Uveitis | Lymph node | Lymphadenitis |
| Nose | Rhinitis | Lymphatic vessels | Lymphangitis |
| Oral cavity | Stomatitis | Appendix | Appendicitis |
| Salivary gland | Sialadenitis | Cecum | Typhlitis |
| Tonsil | Tonsillitis | Rectum | Proctitis |
| Larynx | Laryngitis | Breast | Mastitis |
| Pharynx | Pharyngitis | Testis | Orchitis |
| Lung | Pneumonia | Ovary | Oophoritis |
| Pleura | Pleurisy | Fallopian tube | Salpingitis |
| Liver | Hepatitis | Prostate | Prostatitis |
| Gall bladder | Cholecystitis | Uterus | Endometritis |
| Muscle | Myositis | Urinary Bladder | Cystitis |

Comments (0)