Enzymes (VIva)
Q.1 What are enzymes?
Enzymes are biological catalysts.
Q.2 What is the nature of enzymes?
Enzymes are protein in nature.
Q.3 Name the fastest acting enzyme?
Carbonic anhydrase (CA).
Q.4. What is Km (Michaelis constant)?
Km is defined as that substrate concentration which produces half of the maximum velocity.
Q.5 Km is defined in terms of what?
It is defined in terms of substrate concentration.
Q.6. Which Km value is preferred, low or high?
Low Km value is preferred because at low substrate we can achieve the maximum velocity.
Q.7 Give Michaelis Menten equation?
V1= Vmax [s]
Km+ [s]
Q.8 Some enzymes don’t follow the Michaelis Menten equation then they follow which reaction?
Hill equation
Q.9 Give an example which follows Hill equation?
Hemoglobin
Q.10 Why hemoglobin follows the Hill equation, not Michaelis Menten equation?
Due to the property of heme-heme interaction, hemoglobin does not follow the Michaelis Menten equation as no straight line is followed.
Q.11 Why Linewear-Burk curve or equation is preferred?
Linewear-Burk equation is in the form of a straight line. For a straight line, we need two points, i.e. by selecting a few different concentrations, we can plot the curve and hence find out the Km
Q.12 What are the factors which influence enzyme activity?
Factors which influence enzyme activity are:
- Substrate concentration.
- Enzyme concentration.
- pH.
- Temperature.
- Effect of activators and coenzymes
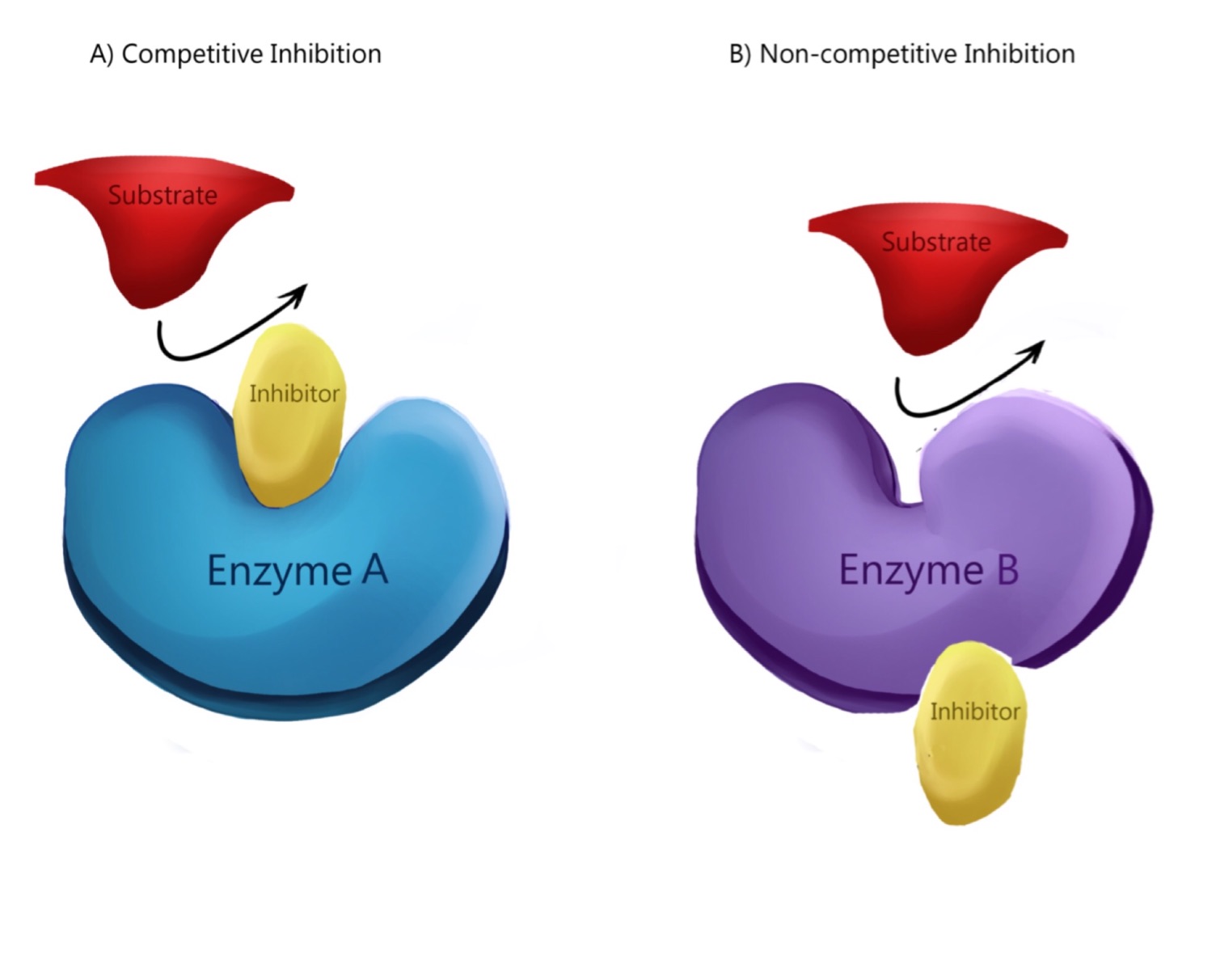
Q.13 What are competitive inhibitors?
Competitive inhibitors are those which has structural similarity with the substrate molecules for the active site of the enzyme.
Q.14 What is the characteristics of line weaver Berk plot in competitive inhibition?
In competitive inhibition, Vmax remains the same but Km value increases.
Q.15 What are the characteristics of lineweaver-Burk plots in non-competitive inhibition?
Reverse of the above is followed in noncompetitive inhibition.
Q.16. What are non-competitive inhibitors?
Non-competitive inhibitors are those which are attached not to the active site but to some other site of the enzyme.
Q.17 Give an example of competitive inhibitors.
Inhibition of succinate dehydrogenase by malonate. L-lactate dehydrogenase (substrate lactate) by oxamate. Cis-aconitase by fluorocitrate.
Q.18 Give the example of non-competitive inhibition.
- Inhibitions of acetylcholine esterase, trypsin, chymotrypsin by diisopropylfluorophosphate.
- Inhibition by heavy metal ions like Ag+ and Hg++ ions.
Q.19. What is the difference between the prosthetic group and the co-enzyme?
- Prosthetic group is tightly bound to the enzyme.
- Whereas co-enzymes exist in the solution in a free state and contact the enzyme only at the site of reaction.
Q.20 What is an allosteric site?
Site other than the active site is called the allosteric site.
Q.21 What are isoenzymes?
Give two examples.
- Isoenzymes are the multiple forms of the same enzymes
- Lactate dehydrogenase,
- Alkaline phosphatase.
Q.22 Name the isoenzymes of LDH?
- LDH has 5 isoenzymes
- HHHH, HHHM, HHMM, HMMM, MMMM.
Q.23 Name the diagnostic enzymes in Ml?
SGOT (AST), LDH
Q.24. Name the diagnostic enzyme in viral hepatitis?
SGPT (ALT)
Q.25 Which enzyme increases in serum in acute pancreatitis?
α Amylase
Q.26. In Wilson disease, which enzyme plays a diagnostic role?
Ceruloplasmin
Q.27 Name the diagnostic enzyme for muscle disorders.
Creatine kinase.
Q.28 Acid phosphatase is a diagnostic enzyme for which disease?
Carcinoma of prostate
Q.29 Enzyme that is a diagnostic tool for bone and liver disease.
Alkaline phosphatase
Q.30 What are anti-enzymes?
Anti-enzymes are the substances produced as a result of repeated injection of certain enzymes in the serum, which prevents the normal action of the enzyme injected.
Q.31 How will you differentiate whether the given reaction is enzyme-catalyzed or not?
We can differentiate the reaction by two tests.
- Heat sensitive test.
- Acid test
Q.32 What are constitutive enzymes?
Constitutive enzymes are those enzymes that are present in constant concentration during the life of the cell.
Q.33 What are inductive enzymes?
Inductive enzymes are those enzymes whose amount present in the cell is variable and depending upon the requirement.
Q.34. What is feedback inhibition?
In a multienzyme reaction, the end-product of the reaction sequence may act as a specific inhibitor of an enzyme at or near the beginning of the sequence with the result the rate of the entire sequence of reactions is determined by the steady-state concentration of the end-product. This type of inhibition is called feedback inhibition.
Q.35 Give an example of feedback inhibition.
Cholesterol inhibits cholesterol synthesis in the liver by inhibition of HMG-CoA reductase.
Also read: Biochemistry Questions & Answers

Comments (0)